Vaccines
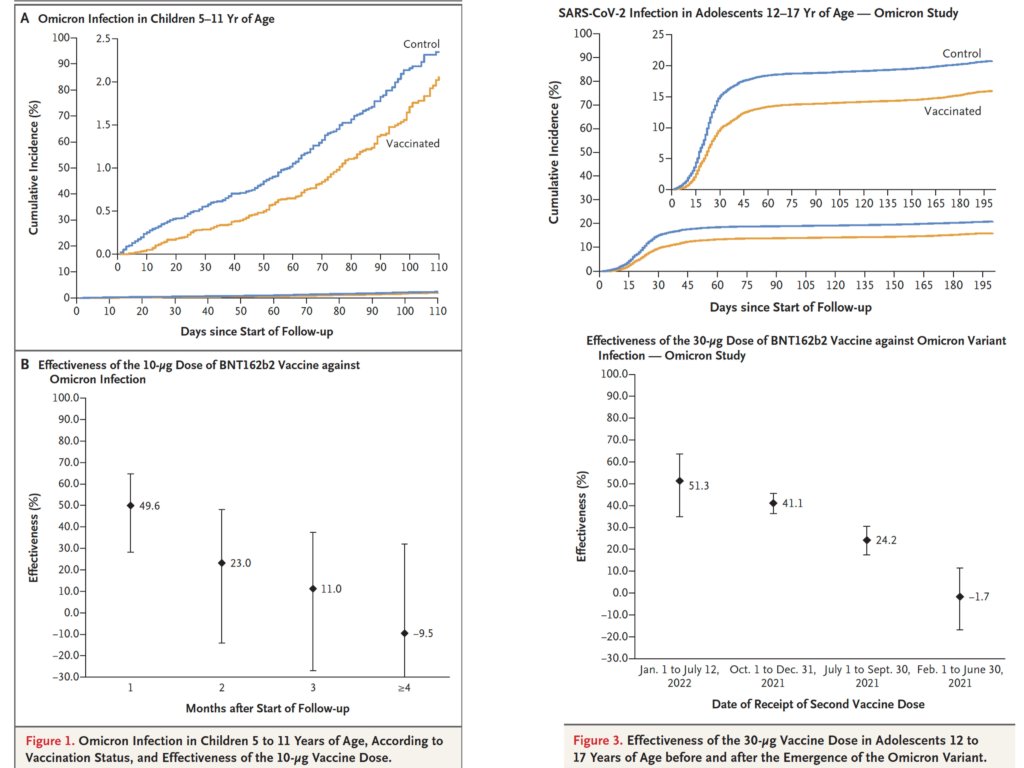
This paper from Qatar says that 2 doses of Pfizer doesn’t actually give kids much protection against Omicron infection, and the protection wanes quite quickly. 🙁
In last week’s General blog post, I gave the disappointing news that two papers found that the bivalent vaccines weren’t that much better than the monovalent boosters. There are three new papers which give confusing and conflicting information.
I dug into this paper from the USA which says that a BA.5 bivalent booster actually does quite a bit better against some of the newer strains than the monovalent booster. (Yay!) It uses a more faithful measuring technique (live virus instead of pseudoviruses), so maybe it is a better study. However, when I dug into the supplemental info of the paper, the data looked slightly flakey. (I choose to believe that this is sloppiness (from trying to get the data out fast) and not malice.)
There were twelve people in each of the three groups (1 dose of monovalent (i.e. original formulation), 2 doses of monovalent, 1 dose BA.5 bivalent) which is kind of small. The groups had significant differences in their composition and treatment. The bivalent group had more prior infections than either monovalent group, was significantly younger (40 years old) than the double-monovalent (65.7 years old), and the serum collection was sooner on average since the last booster (22 days) than the double-monovalent group (82.7 days).
The differences between the single-monovalent and the single-bivalent groups were not as stark: the average time between booster and collection for the single-monovalent samples was 21 days (although all were either 1 week or 4 weeks; the standard deviation of the bivalent group was much lower); the average age for the single-monovalent group was 33 years old.
I thus think that the improvement in antibody levels going from the double-monovalent to the single-bivalent is probably an overestimate. However, the difference between the single-monovalent and the single-bivalent probably is meaningful. In this small study, one dose of the monovalent (where 2/3 of the group got Pfizer, 1/3 Moderna) was not as good as one dose of the bivalent (with 1/4 Moderna, 1/12 Pfizer, and 2/3 unreported).
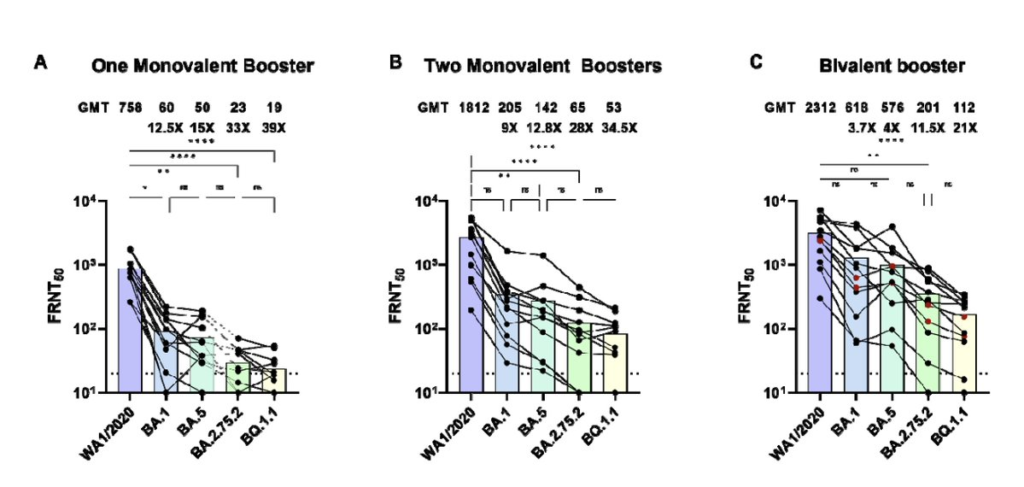
This paper from the USA says that one booster of bivalent BA.5 is better than one booster of monovalent, but they are both pretty shit against XBB.1:

This paper from Israel, like last week’s papers, says that the bivalent is not much better than the monovalent:

This preprint from Qatar says that people with three doses were more likely to get reinfected with an Omicron infection that those who only had two doses. (Two or three doses was still better than zero doses.)
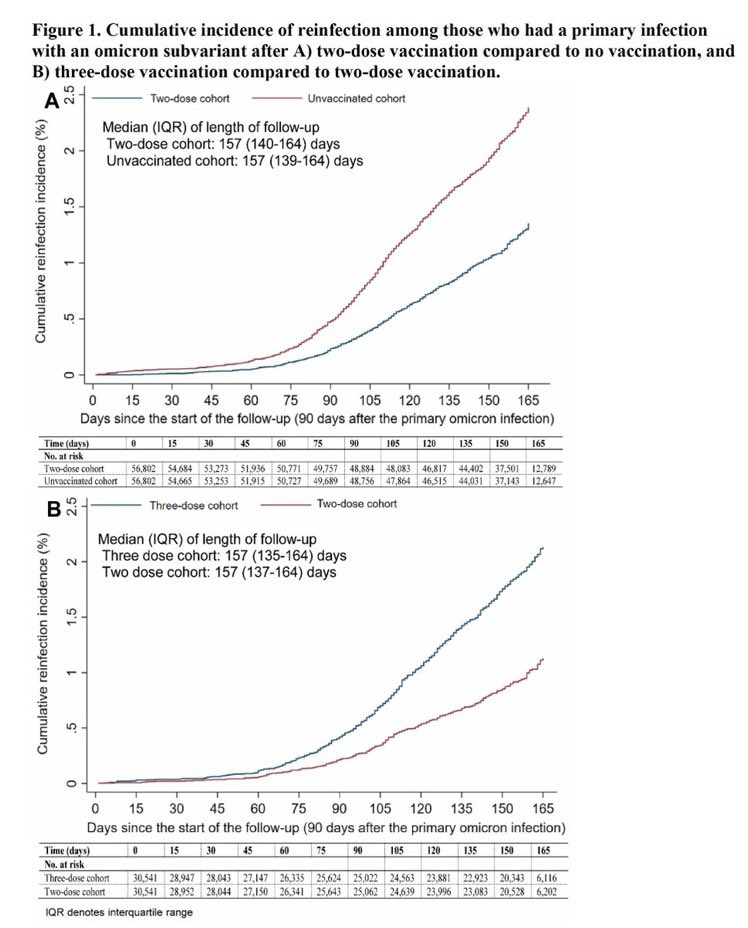
This is a surprising result, and suggests that maybe there is some imprinting going on.
Also, I keep thinking about the US CDC graph (from this page) which shows that one booster (solid dark blue) doesn’t give much survival advantage over two (dashed light blue), while two boosters (solid purple) really does, at least in people over 50:

(It might be, however, that people who get double-boosted are just more careful than the single-boost people.)
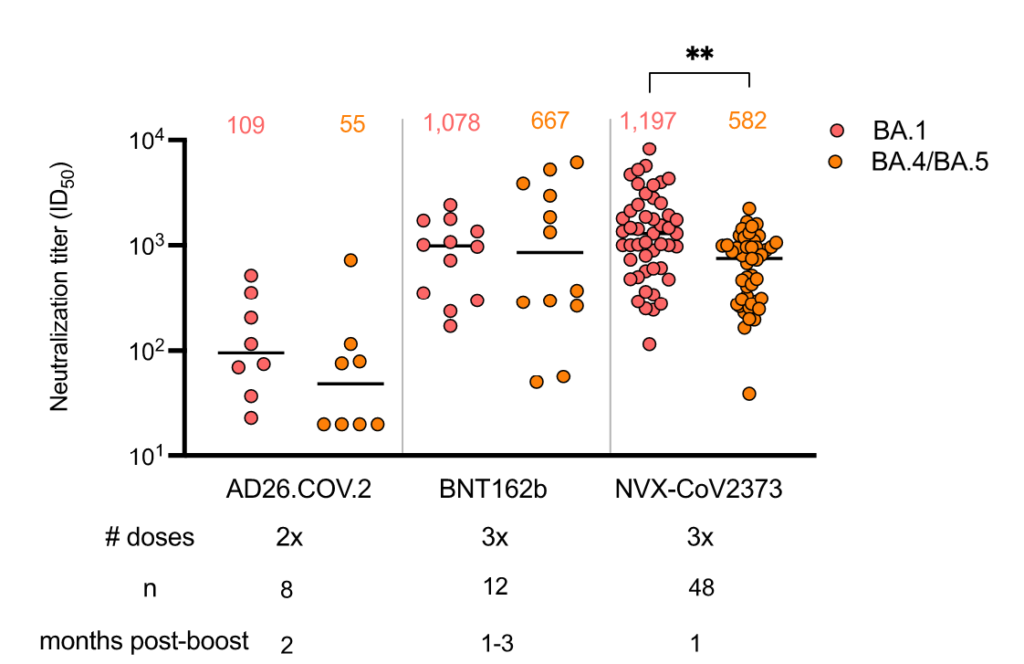
This preprint from South Africa says that Novavax doesn’t do as well against BA.1 or BA.5 as it does against earlier strains (no surprise). It says that Novavax’ effectiveness is comparable to mRNAs and better than AZ:
This paper from Germany says that boosters bump up antibody levels but not the number or effectiveness of T-cells.
Pathology
This tweet thread interprets this report from Australia on excess deaths. (Yes, yes, it’s Australia and not Canada, but I suspect there are things that are common between the two countries.) There were a ton of excess deaths in 2022 — +16% in July 2022:
Some were listed as COVID-19, but a ton of the excess deaths came from heart problems of various types:
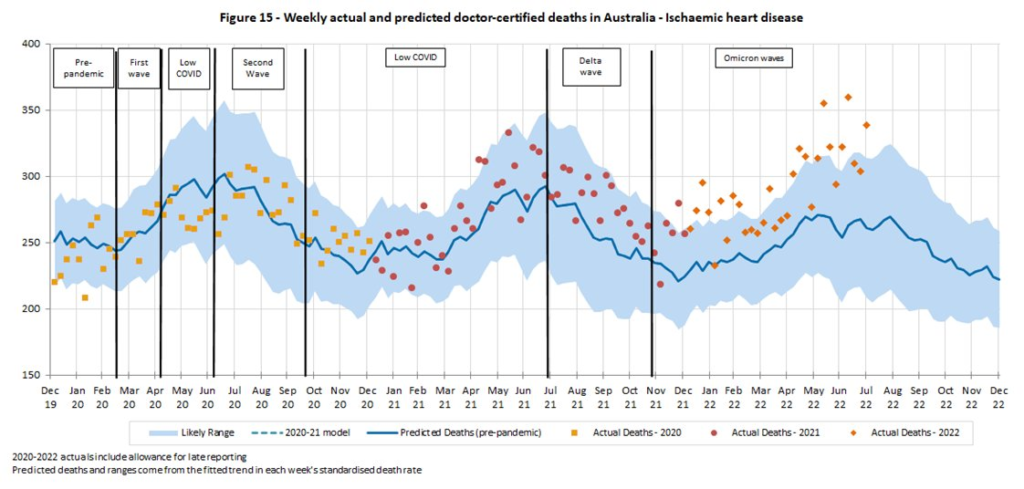
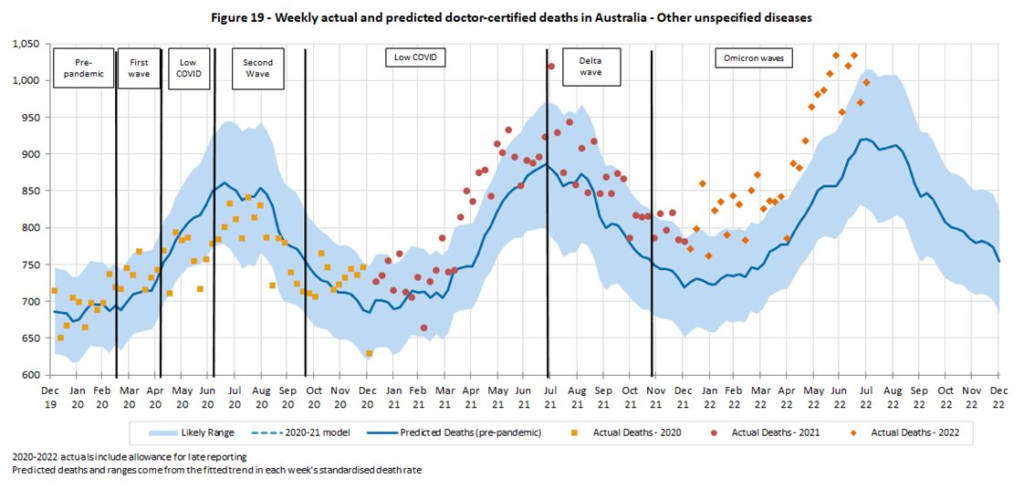
and unassigned causes of death:
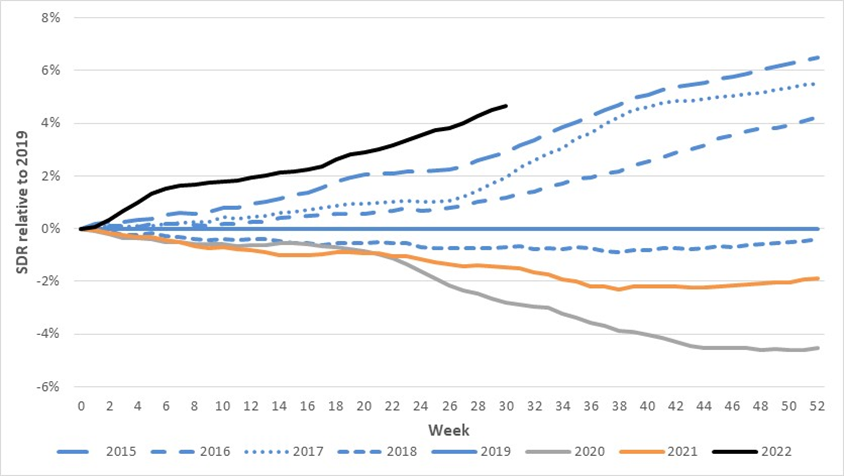
This graph of excess deaths by year relative to 2019 shows that deaths are way higher in Australia than they were in 2020 and 2021:
This paper from the USA says that the vast majority of COVID-19 patients develop antibodies to PF4 (a totally normal element of human blood which inhibits clotting), and the level of anti-PF4 antibodies correlates with the severity of the acute disease.
The alert reader will recognize PF4 being implicated in the (rare) clotting problems that the AZ vaccine caused. This paper, for example, says that the adenovirus used in the AZ vaccine can bind to PF4 — acting much like an anti-PF4 antibody.
I thought about putting this in the Long COVID section because while it is not quite a smoking gun Long COVID, it is certainly suspicious. (One of the leading theories on what causes Long COVID is micro-clots.)
This paper from the USA says that a combination of antibiotics and COVID-19 can mess up your gut biome, and that it can help microbes from your gut get loose into your bloodstream. It’s not clear how much is from COVID-19 and how much from antibiotics, though this tweet says that they see some leaky gut in COVID-19-infected mice (who did not get antibiotics).
This paper from the UK found a few genes which made people more susceptible to COVID-19. These genes were also associated with lupus. Some of the genes which had similar effects for lupus and COVID-19 (i.e. were correlated), but there were some genes which had opposite effects for lupus and COVID-19 (i.e. were anti-correlated).
This paper with an international team looked at genetics of people all over the world, and found that toll-like receptor TLR7 (on the X chromosome) was associated with a 5.3-fold increase in severe disease, and was not sex-linked.
This paper from Mexico (from January 2022) found that probiotics reduced viral COVID-19 loads, infiltrates and the duration of both digestive and non-digestive symptoms!
Treatments
This paper from Belgiam looked at using throat sprays of lactobacillaceae (frequently used in probiotics) shortly after COVID-19 infection. The study was small, self-selected, and partially survey-based, but there seemed to be small but significant benefit from the throat spray. Symptom severity seemed to be inversely correlated with the amount of lactobacillaceae measured in the throat; 6.7% of the treated group tested positive after three weeks, versus 26% of the placebo group. This study was by no means a slam dunk, but I think it hints at interesting future explorations.
Long COVID
There are two books for long haulers which just came out. I haven’t read either book, so I can’t recommend (or dis-recommend), but if you or someone you care about has Long COVID, maybe they will like one of these books. (Let me know what you/they think!)
This paper from Finland gave remdesivir to hospitalized patients and followed up a year later. They found that whether you got remdesivir or not did not change the number of people with Long COVID after a year. About 15% in the remdesivir group and 15% in the standard care group had Long COVID a year later.
Recommended Reading
This long general-audience article is on what’s up with nasal vaccines.
This article talks about prophylactic nasal treatments.
This article talks about how Evusheld — a monoclonal antibody that immunocompromized people could take to functionally act sort of like a vaccine — doesn’t work against the new variants. Ulp.
This article from the USA talks about the crisis they are having at pediatric hospitals with flu, COVID-19, RSV, rhinoviruses, and enteroviruses. Is Canada facing the same? I don’t know.
