Vaccines
This study in Sweden of mix&match effectiveness against symptomatic infection found that a second dose of mRNA was better after a first of AZ than a second dose of AZ:
| 1st Dose | 2nd Dose | Effectiveness |
| AZ | AZ | 50% |
| AZ | Pfizer | 67% |
| AZ | Moderna | 79% |
| Pfizer | Pfizer | 78% |
| Moderna | Moderna | 87% |
This article says that Pfizer has requested authorization from Health Canada for its pediatric vaccine. (Funny, this article said so on Friday.) However, the new article had a piece of info I hadn’t seen: that the Government of Canada signed a contract for pediatric vaccine “last spring” (which I assume was Spring 2021).
This article says that the vaccine from Valneva, a French pharma company, did really well in its Phase 3 trials, and are requesting approval from Scottish authorities. (It’s made in Scotland.)
It’s difficult to say exactly what the efficacy was. Because there are now effective vaccines, it is unethical to run trials which have new-vax vs. placebo; you need to run trials of new-vax vs. old-vax. (These kinds of studies are called “non-inferiority” studies.) Valneva set up their study to show non-inferiority to AZ… but in the middle of the study, they stopped giving AZ to young people because of blood clots.
Still, they were able to prove that it’s not worse than AZ, and they think it’s better.
This preprint looked at AZ, Pfizer, and Moderna over time:
| Vaccine | after 32 days | after 4 months |
| Moderna | 87% | 73% |
| Pfizer | 85% | 58% |
| AZ | 65% | 32% |
The paper found no clear evidence that effectiveness changed when Delta took over.
This preprint looked at waning. There were some steep drops (especially for J&J) in effectiveness against infection, but those were much much smaller in effectiveness against hospitalization.
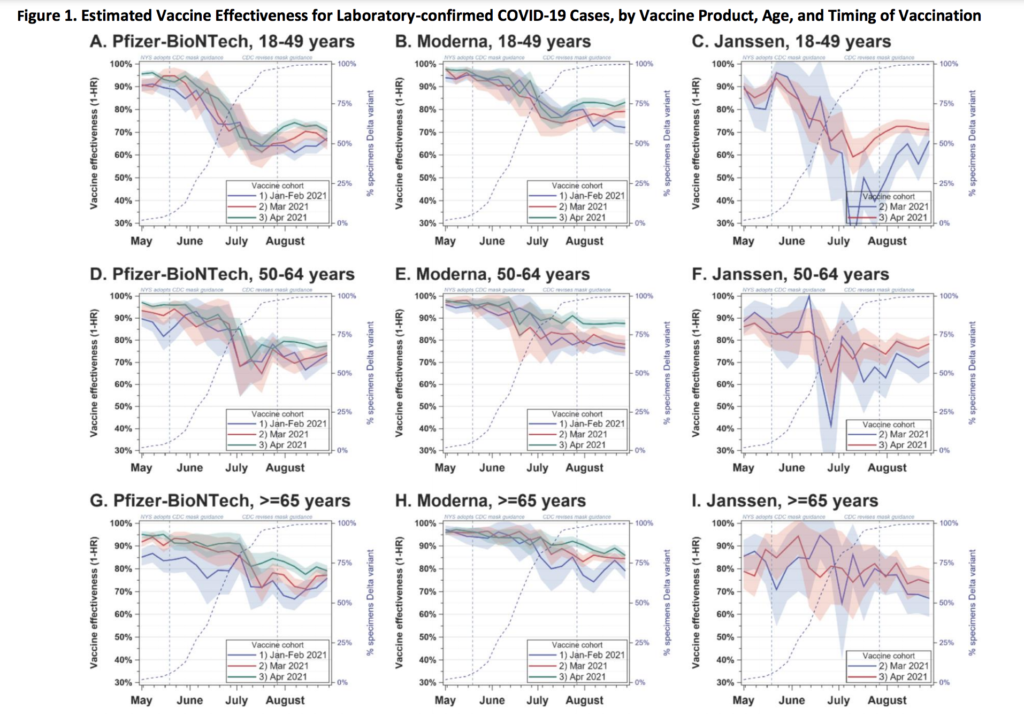
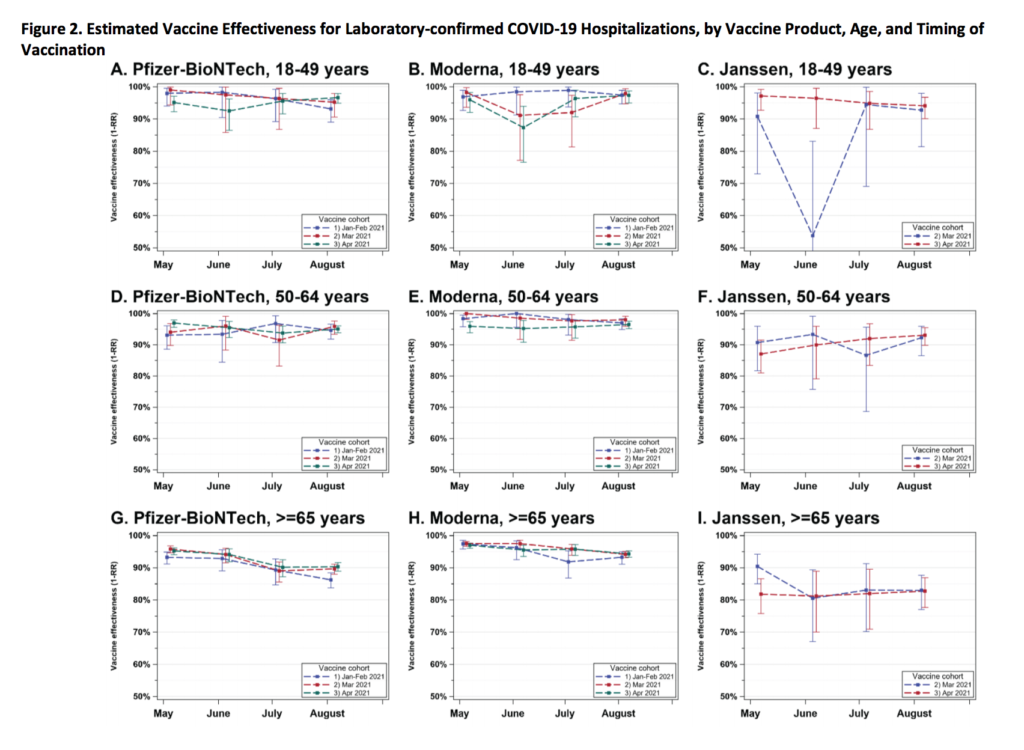
Mitigation Measures
This article says that the Canada Emergency Rent Subsidy (CERS) and the Canada Emergency Wage Subsidy (CEWS), the Canada Recovery Benefit (CRB), the Canada Recovery Sickness Benefit (CRSB) and the Canada Recovery Caregiver Benefit (CRCB) are set to terminate on Saturday, 23 October. The government could extend them through 30 November easily, but to extend them past that would take a new act of Parliament.
